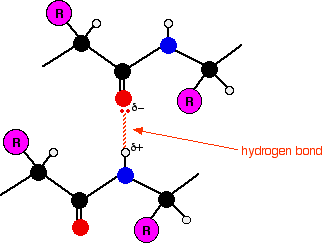
Thus, a more efficient computational method is desired. It has been shown that the BSSE correction of the potential energy surface leads to sizeable differences in the H-bond lengths for correlated calculations using split-valence basis sets with polarization and diffuse functions.Īlthough much progress has been made for investigating the nature of H-bonds utilizing quantum chemistry methods, few ab initio calculations have been carried out on the calculation of dynamic properties of hydrogen bond interaction for biomolecules due to relatively large size of systems. Therefore, BSSE introduces a nonphysical attraction between the two monomers. Applying the basis set of the complex to each molecule will yield a lower energy than that from the calculation with the basis set for the monomer itself. The basis set superposition error (BSSE) is another issue to be considered in the computation of H-bond complexes, which arises because of the incompleteness of the basis set functions. have shown that in the case of some systems with weak H-bond such as C-H π or O-H π, B3LYP does not offer the same level of reliability as MP2. It has been shown that B3LYP calculation provides very good description of strong H-bond systems, comparing with the experimental data or MP2 results. Density functional theory (DFT) methods such as B3LYP is becoming more and more popular these days. However, even a single point calculation at MP2 level of medium-sized hydrogen bond systems can be very demanding. Thus, Møller–Plesset methods such as MP2 can be considered ideal for the study of hydrogen bonds. Molecular orbital methods, especially the post Hartree–Fock (HF) methods, are capable of providing an accurate description of systems containing hydrogen bonds. Quantum chemistry method is an ideal approach to calculate the energy of a hydrogen bonded system since all electronic and steric effects are fully taken into account. The formation of an H-bond can usually cause red shift of IR spectroscopic bands, even though improper blue shifts have been also detected. Now, it is well accepted that, in neutral systems, H-bonds formed between O, N, or O/N pairs of atoms are very strong, whereas those formed between C-H and a π system are weak. In practice, a cutoff of 120 or 90º is employed. Furthermore, the D–H–A angle should be close to 180º. When a hydrogen bond is formed, the distance between the H and the acceptor (A) atoms has to be smaller than the sum of their corresponding van der Waals radii. The geometry of the H-bond complex, and in particular, the distance and angle around the H-bond also provide a very good indication of such interaction. The interaction energy is the best indicator of H-bond strength. There exist many hydrogen bond donors and acceptors in proteins, such as the amide and carbonyl groups of the backbone, as well as the polar functional groups (amides, carboxyl groups, hydroxyl groups, and amines) in the side chains of some amino acids.ĭifferent parameters can shed light on the different aspects of the nature of H-bonds established for a given system. Hydrogen bond is formed when an electronegative atom (H-bond acceptor, A) approaches a hydrogen atom bonded to another electronegative atom (H-bond donor, D). Therefore, correctly describing the thermodynamic properties of inter/intra-protein hydrogen bonds is essential. α helix and pleated β -sheet were predicted in 1951 by Linus Pauling and Robert Corey on the basis of hydrogen bonding and cooperative criteria. They play an indispensable role in the stabilization of the native structure of a protein. Hydrogen bonds, providing most of the directional interactions, are the dominant feature in the formation of protein secondary or tertiary structures. During folding process, the unfolded structure collapses first to minimize the area of hydrophobic surface that is exposed to the solvent and hydrogen bonds begin to form. The native structure of protein at physiological temperature and solvent condition, which is located at a free energy minimum, is a result of delicate balance between many competing interactions, including van der Waals forces, hydrophobic interaction, and electrostatic interaction, such as salt bridge and hydrogen bond (H-bond). The function and chemical properties of protein are determined by its three-dimensional structure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
